Identifying patients possibly infected with COVID-19 and rapid testing are key in containing the spread of the virus. At the same time, the pandemic also created a severe nationwide shortage in NP swabs used to collect specimens from patients.
To address this new crisis, together we are producing 3D printed flexible swabs for immediate distribution. This swab design not only provides better accuracy than the current COVID-19 testing standard, but meets the demand for more testing. We are currently printing 20,000 swabs a day from our print farms, which is a fraction of our full capacity.
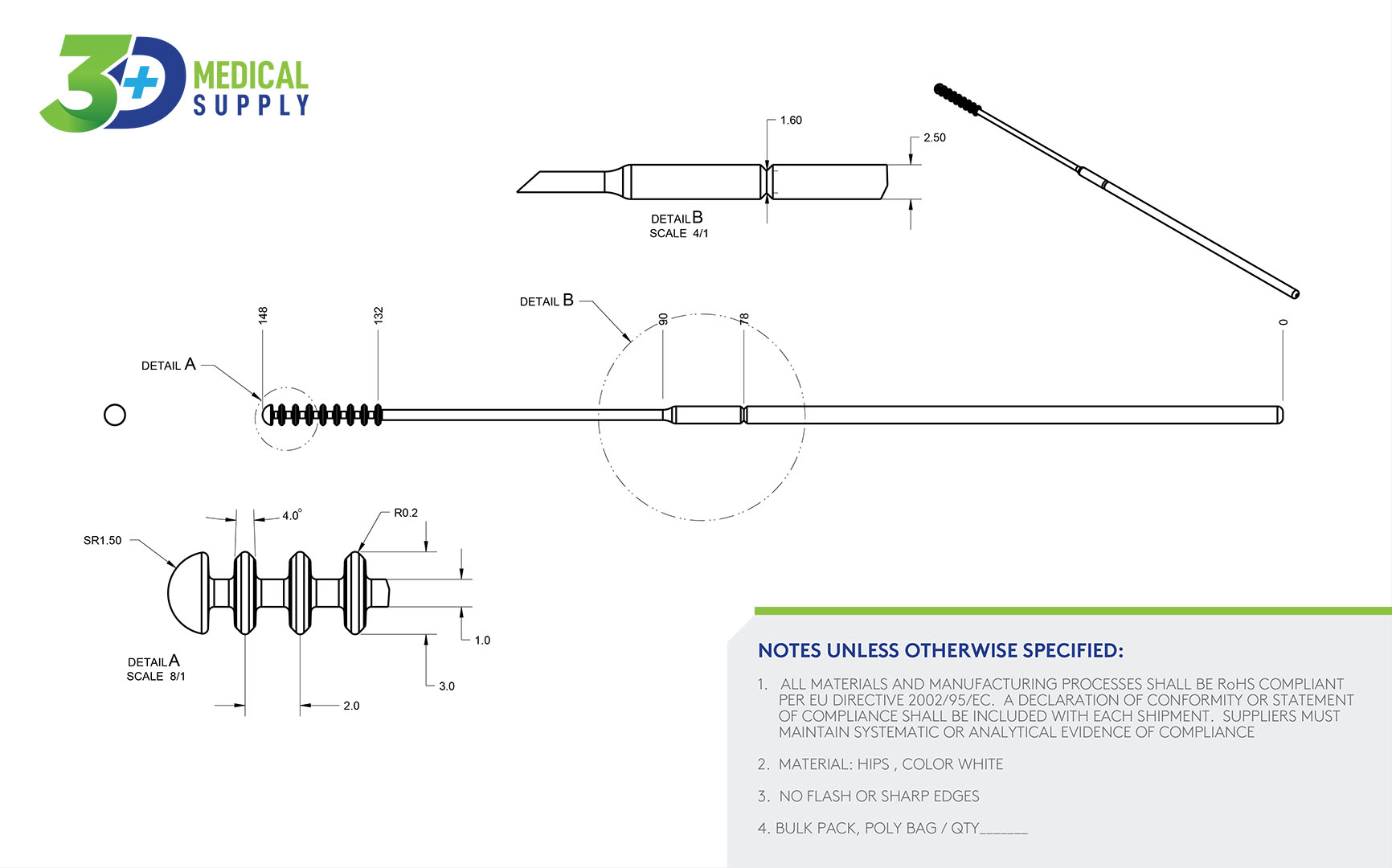
The NPSFlex™ Swab
The key component of the NPSFlex™ is its versatility and use of Injection molding and Polypropylene (PP) material. The swab is flexible enough to enter the nasal cavity and designed with durability.
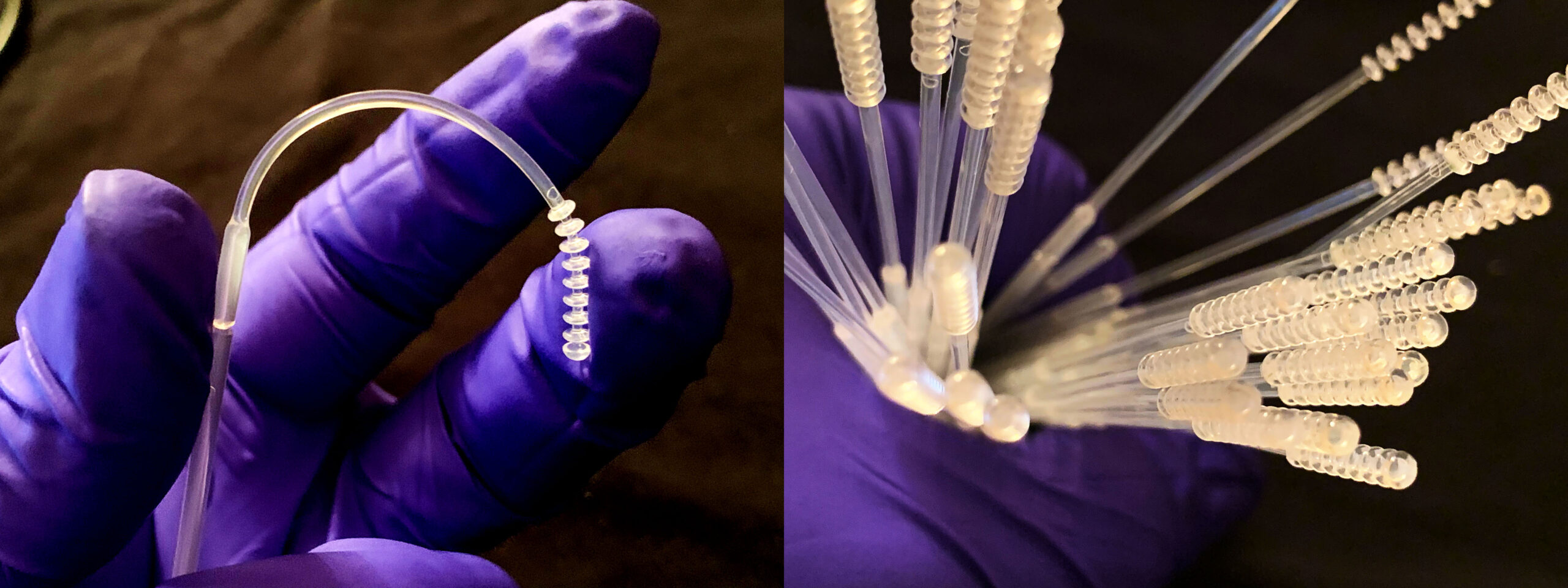
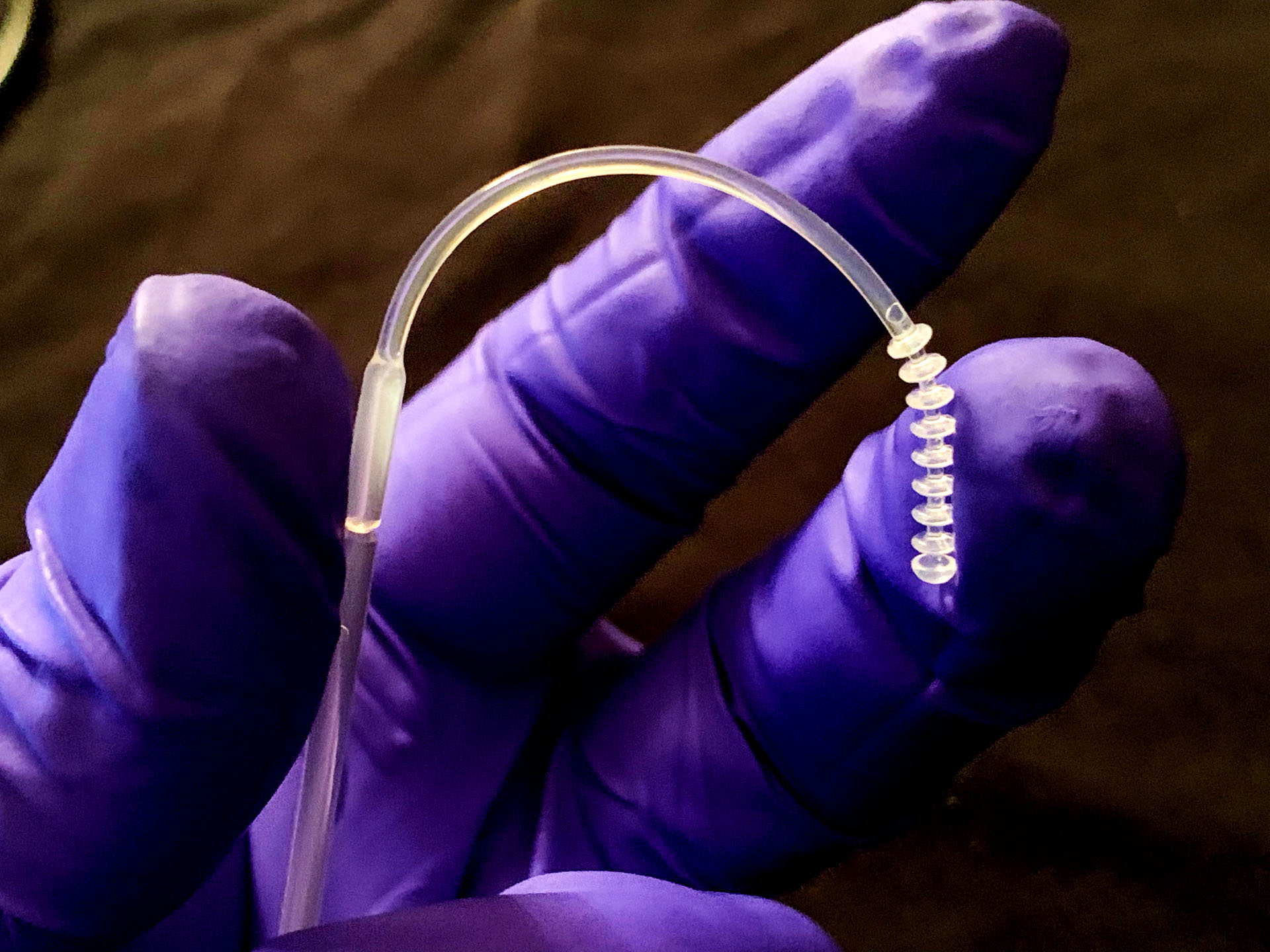


NPS Flex Testing Swab Reaches New Dimension with Injection Molding
Increases tests to up to 10 million per month
To meet the growing demands for Covid-19 testing, 3D Printing LLC in partnership with 3D Medical Supply have printed a tool for injection molding technology to meet high volume output of swabs with accurate results. The collaboration between the two companies offers technology testing solutions to assist local communities during this global pandemic.
Injection molding essentially replaces conventional swabs that use flocked fibers and are made on specialized, proprietary equipment in limited supply. The demand for test swabs for both nasal and throat swab tests will continue to grow due to Covid-19.
Benefits of Using Injection Molding
- Swabs are made with Polypropylene (PP), a widely used thermoplastic valued for its durability, elasticity, transmissivity, and insulating properties used for a multiple purposed including for medical components.
- Have a stable thermoplastic handle and a head made of a thermoplastic elastomer to make the examination less uncomfortable for the patient.
- Deliver high product quality with high-volume output and great cost-effectiveness.
- 32-cavity mold and a cycle time of 6 to 8 seconds, processors can achieve an output of up to 320 test swabs per minute and 460,000 test swabs per day with a single production unit.
- Highest precision with the shortest cycle times
CHARACTERISTICS OF SPRO12W
- Good chemical resistance
- Excellent surface resolution and feature details
- Low moisture absorption
- Produce durable production parts without tooling
NPSFlex™ Specifications
The NPSFlex™ swabs are individually packaged in autoclavable bags,
and are very resilient to high temperatures with a melting temperature of 175°C. This allows them to be sterilized by using an AutoClave, Ethylene Oxide (EtO), or Gamma Radiation sterilization process.
Frequently Asked Questions
Is the NPSFlex™ swab FDA Registered?
Yes, the FDA Registration number is 3016977282
Why use Injection Molding?
Injection molding is the better choice for testing because it provides a better surface finish and quality in parts. 3D printing alone, on the other hand is also be beneficial for low-volume production, but fabrication of large quantities may not always be realistic—especially in comparison to injection molding.
What’s the difference in the testing process?
An injection molding swab is inserted through the nose into the back of the throat and rotated to collect the patient’s RNA. The RNA is then tested in a lab for the presence of COVID-19.
Does the manufacturer have an FDA Owner/Operator number?
Yes, the FDA Owner/Operator number is 10075173.
What is the manufacturer’s capacity?
Up to 100,000 a week per print farm (which is a fraction of our full capacity).
Are the swabs individually packaged and sterilized?
Depending on the clients’ needs, the swabs can be individually packaged and sterilized or individually packaged and non-sterilized.
Is there a minimum order amount?
Yes. 5,000 swabs are the minimum order amount.
The NPSFlex™ Swab Advantage
- Other swabs in the marketplace are glasslike and can break. The NPSFlex™ swab is both flexible and has high tensile strength to allow the clinician easy and safe access to glide through the nostril, along the septum floor, to the nasophyrangeal wall with no risk of breakage.
- NPSFlex™ swab is smooth to the touch and provides ultimate patient comfort in the testing experience.
- NPSFlex™ is manufactured in the USA which eliminates worries of shortages due to export limitations from China or import red tape at ports.
- NPSFlex™ is a reliable and superior product made to exceed the current standard during this time of critical need.
| SWAB IDENTIFICATION | |
| PRODUCT NAME | sPro12W |
| MATERIAL NAME | Polypropylene (PP) |
| APPLICATION | – Packing material – Bag in pharmaceutical – Housings parts – Oil tube, connector – Sport and leisure goods |
| FOR USE WITH | MfgPro2 – 30 xS |
| SPECIFICATION | |
| COLOR | White |
| DENSITY | 0.96 g/cm3 |
| PACKING DENSITY | 0.43 g/cm3 |
| MECHANICAL PROPERTIES | ||
| Tensile Strength (Mpa) | Tensile Modulus (Mpa) | Elongation |
| 49 | 1593 | 11% |
| Flexural Strength (Mpa) | Flexural Modulus (Mpa) | Impact (KJ/m2) |
| 82 | 1623 | 2.6 |
| THERMAL PROPERTIES | |
| Melt Temperature | Operation Temperature |
| 175°C | 70°C |